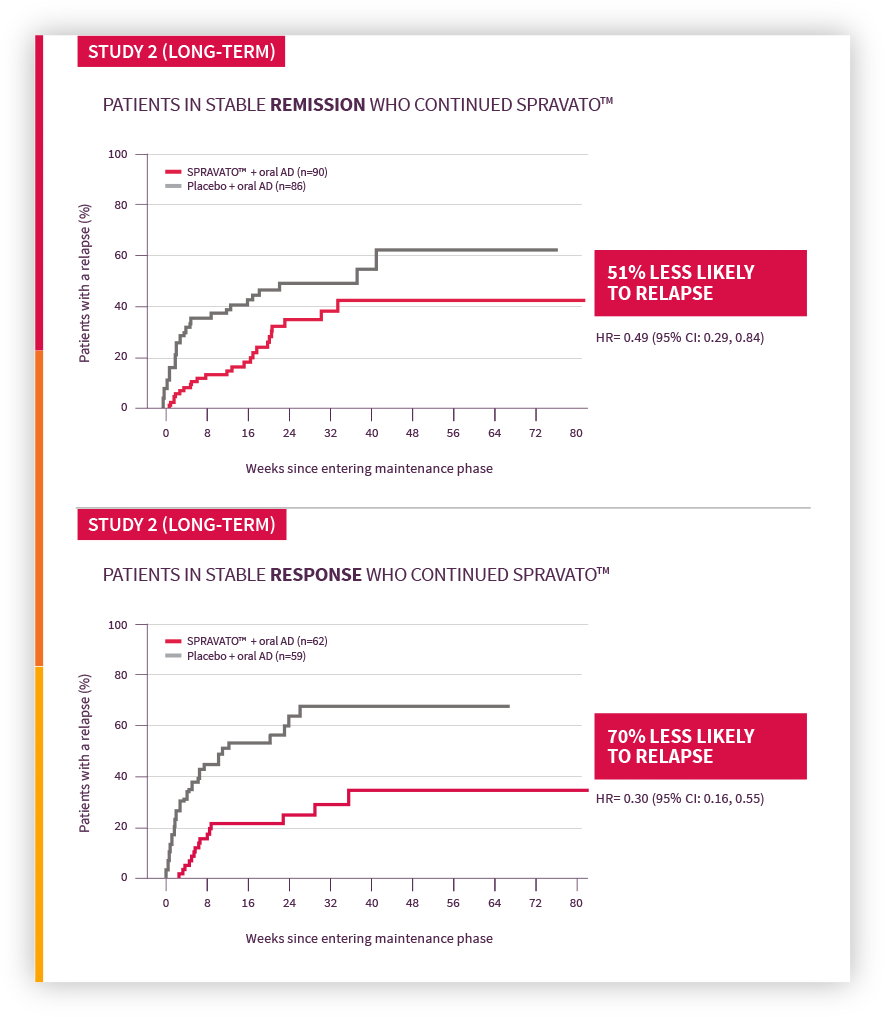
Concomitant Use of SPRAVATO With Antidepressant Monotherapy or Augmentation Therapy in Clinical Trials in Major Depressive Disorder and Suicidal Ideation with Intent

J&J scores Spravato trial win in high-risk depression. Will doctors and payers buy in? | Fierce Pharma

Janssen Announces U.S. FDA Approval of SPRAVATO™ (esketamine) CIII Nasal Spray for Adults with Treatment-Resistant Depression (TRD) Who Have Cycled Through Multiple Treatments Without Relief

Janssen Announces U.S. FDA Approval of SPRAVATO® (esketamine) CIII Nasal Spray to Treat Depressive Symptoms in Adults with Major Depressive Disorder with Acute Suicidal Ideation or Behavior
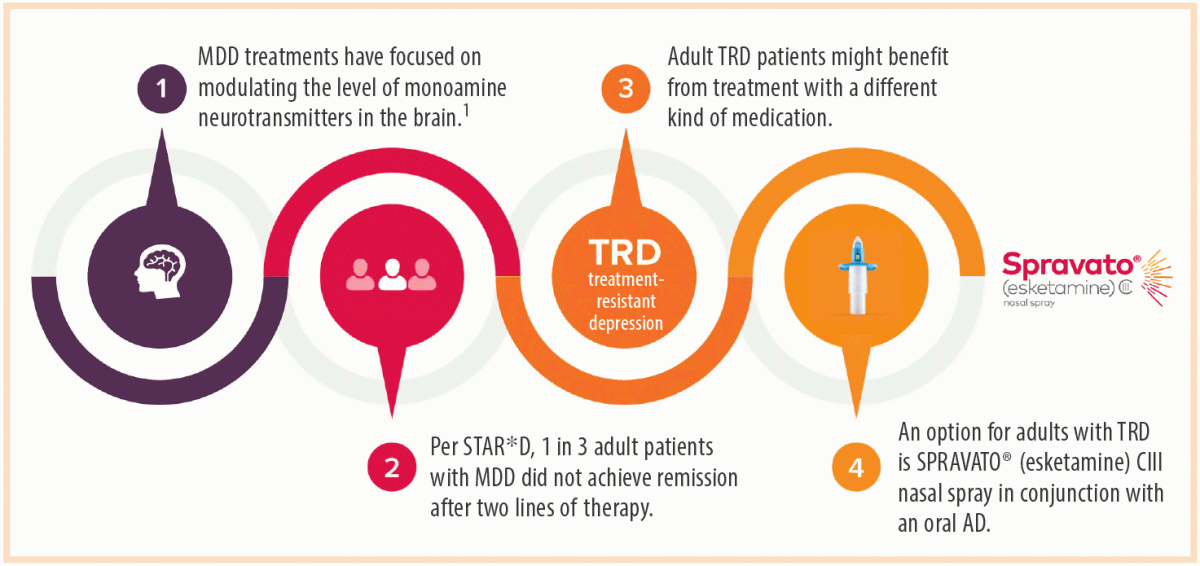
SPRAVATO® (esketamine) CIII Nasal Spray: The First and Only N-Methyl D-Aspartate (NMDA) Receptor Antagonist Approved in Conjunction With an Oral Antidepressant for the Treatment of Adults With Treatment-Resistant Depression | Psychiatrist.com

Spravato 28 mg nasal spray, solution - Summary of Product Characteristics (SmPC) - print friendly - (emc)