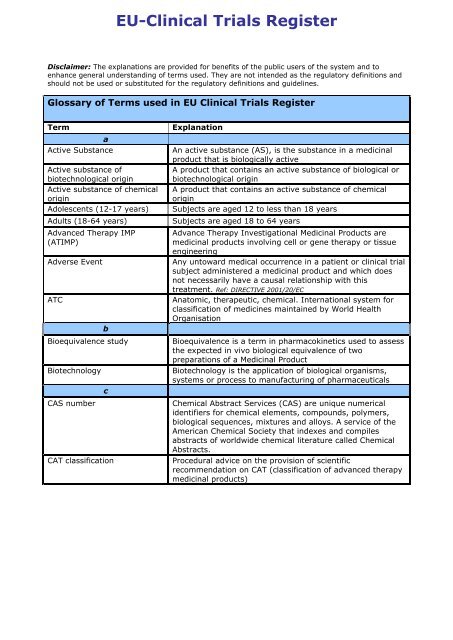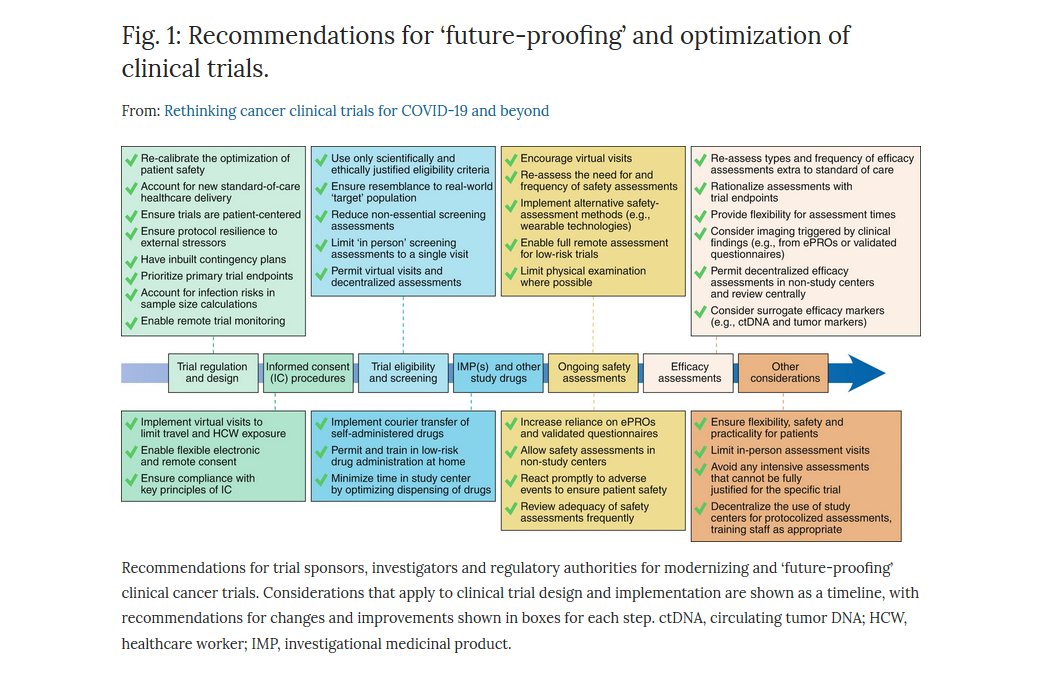
Nature Cancer on Twitter: "ONLINE NOW @NatureCancer: "Rethinking cancer clinical trials for COVID-19 and beyond" by @GaryJDoherty A great discussion about lines of action to ensure the resilience of clinical #cancer research

Trial summary and protocol for a phase II randomised placebo-controlled double-blinded trial of Interleukin 1 blockade in Acute Severe Colitis: the IASO trial. - Abstract - Europe PMC

Proposed clinical trial application dossier. AMPs auxiliary medicinal... | Download Scientific Diagram
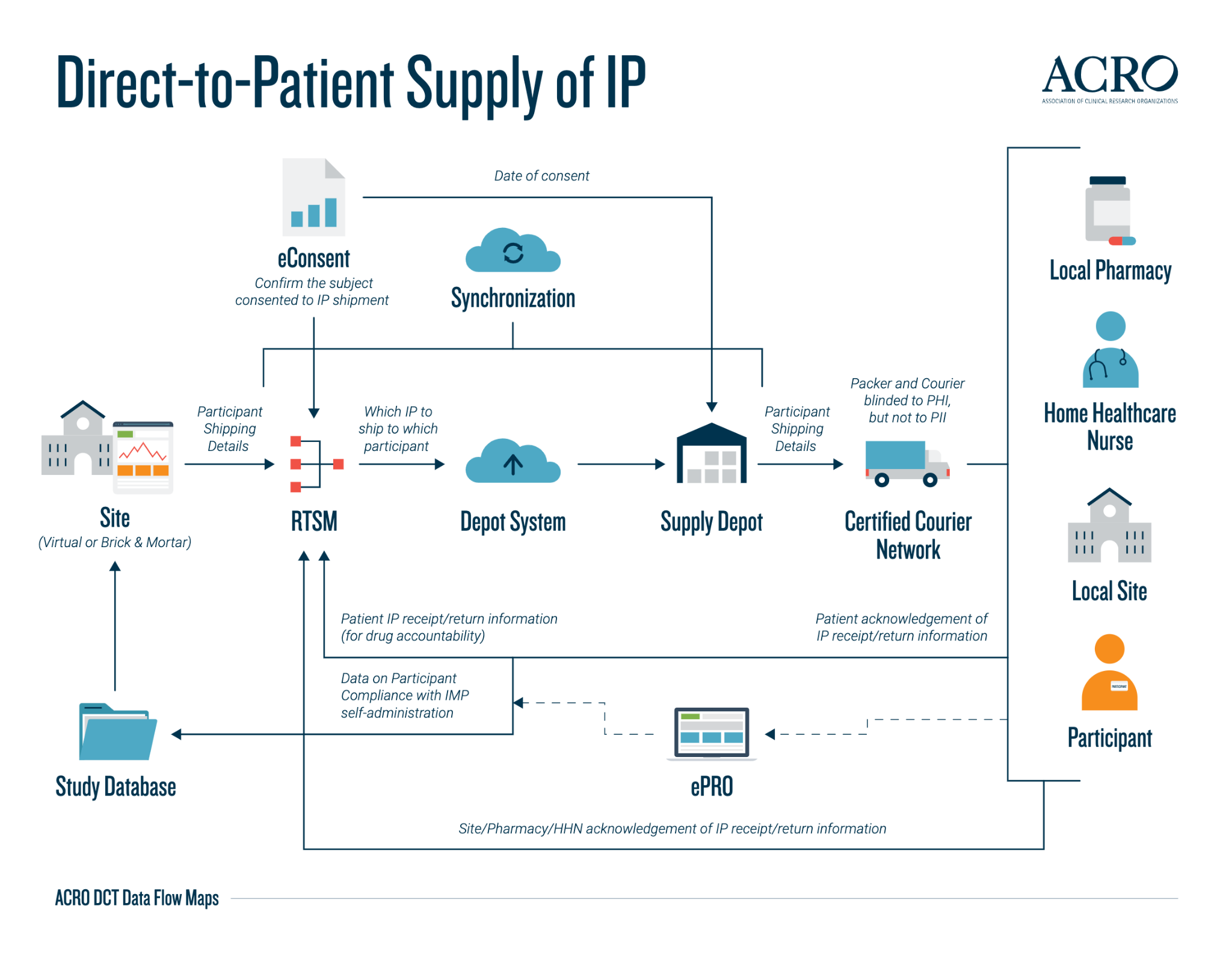
DCT(分散型臨床試験)データのフローマップ | Association of Clinical Research Organizations | Medidata Solutions - Medidata Solutions