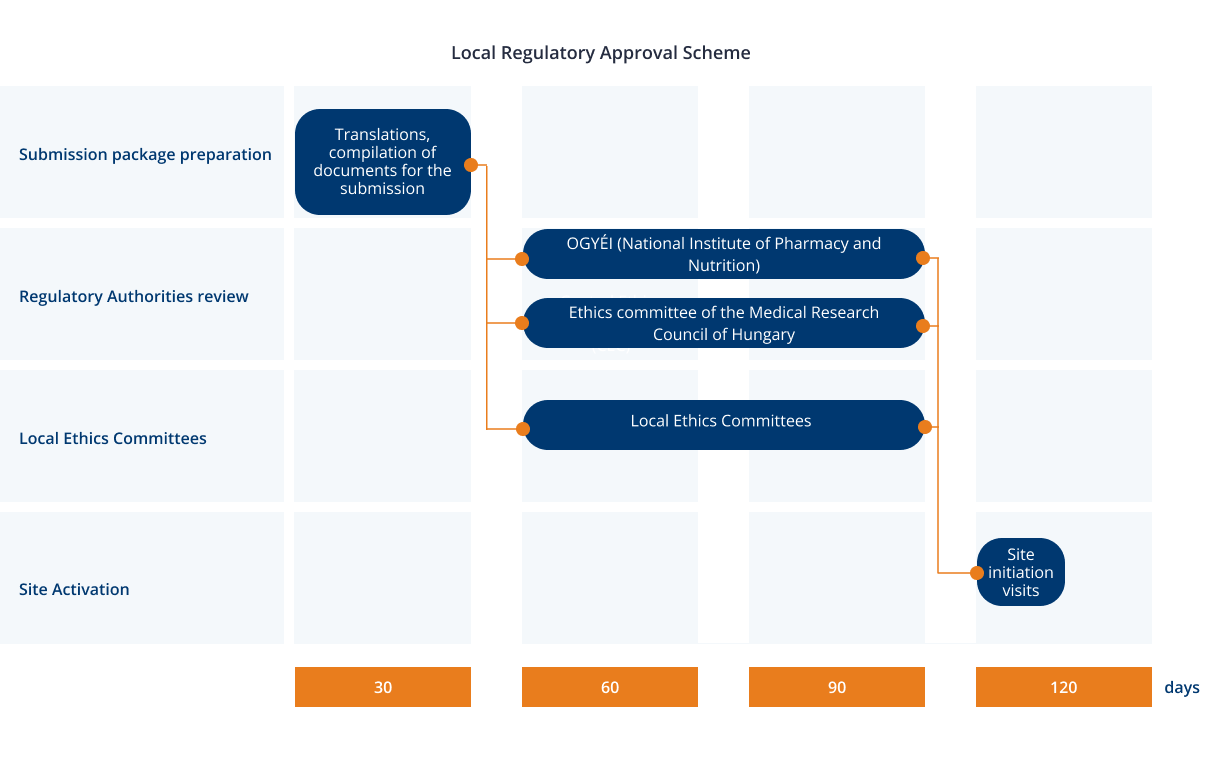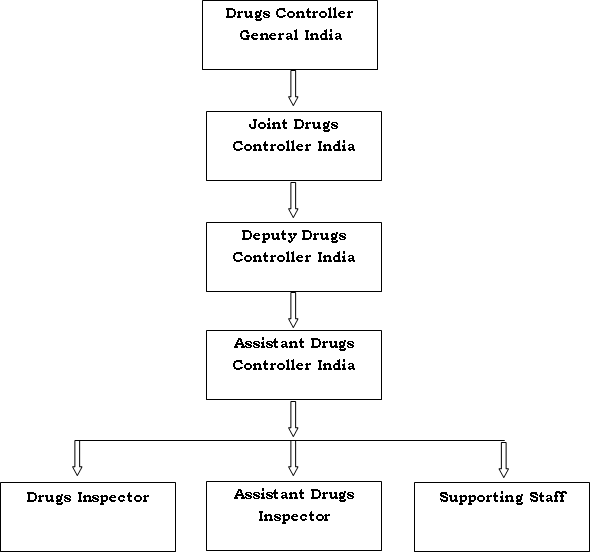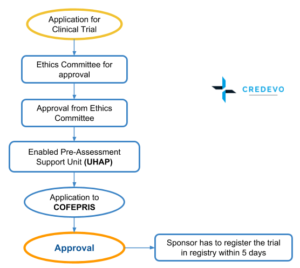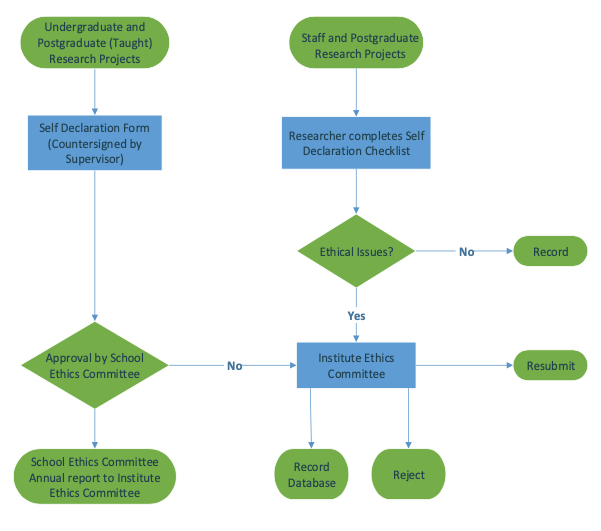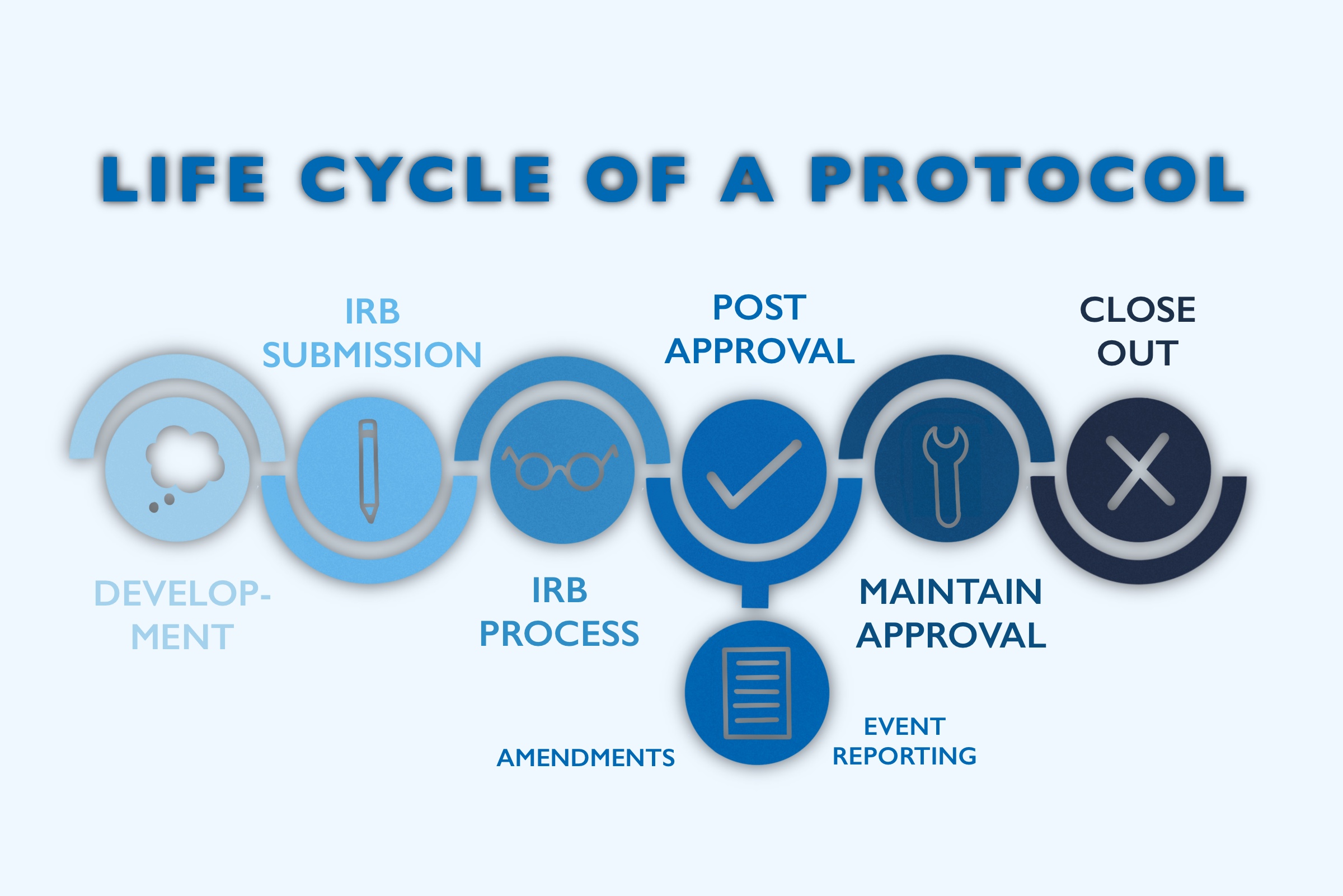
How do Institutional Review Boards (IRB) and Ethics Committees (EC) impact clinical trials? - Clincierge
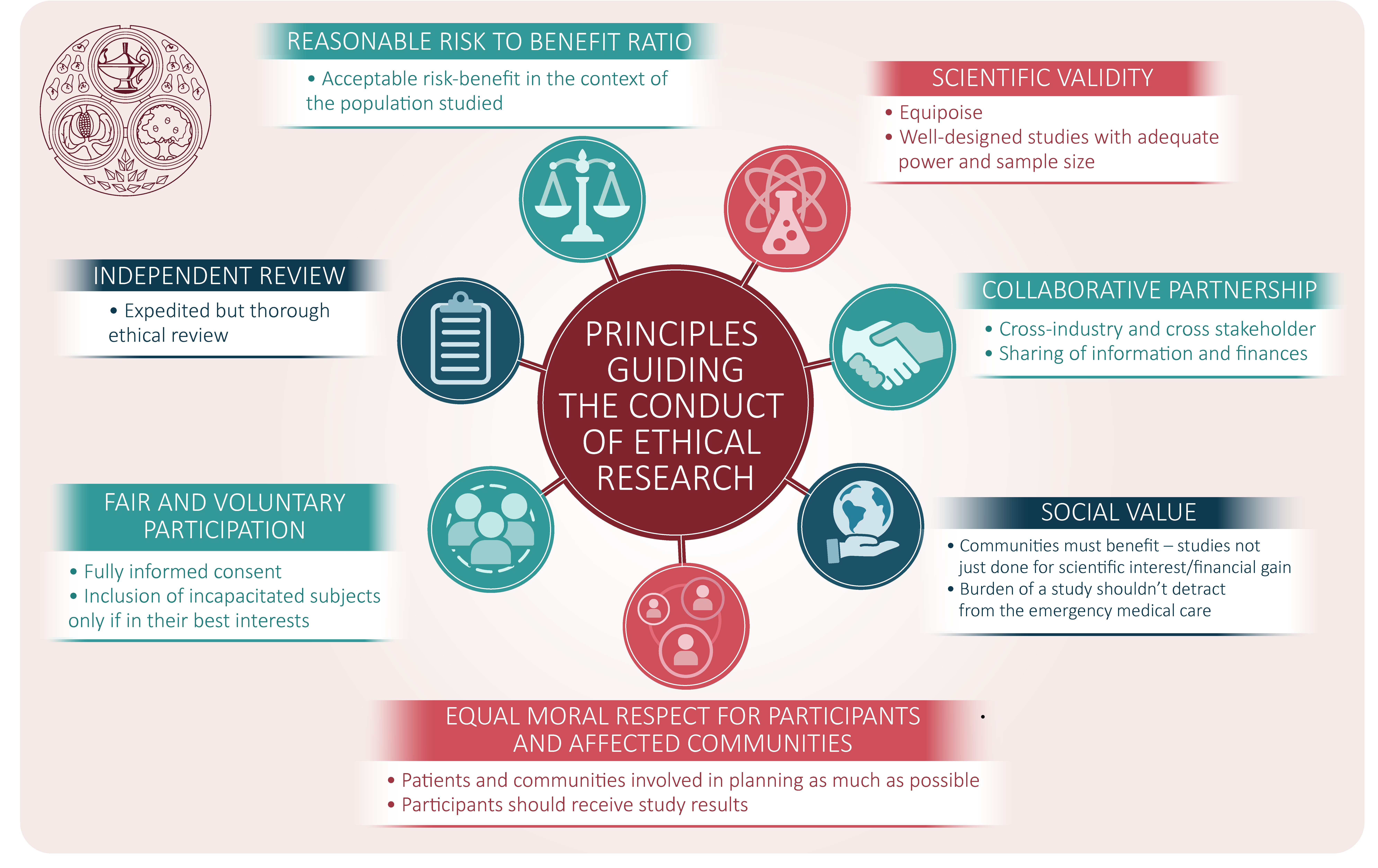
The ethics of conducting clinical trials in the search for treatments and vaccines against COVID-19 - FPM

Oct 06 Ethical and Regulatory Approval Process in HIV Vaccine Clinical Research India Experience HIV Vaccine Clinical Research Ethical and Regulatory Issues. - ppt download