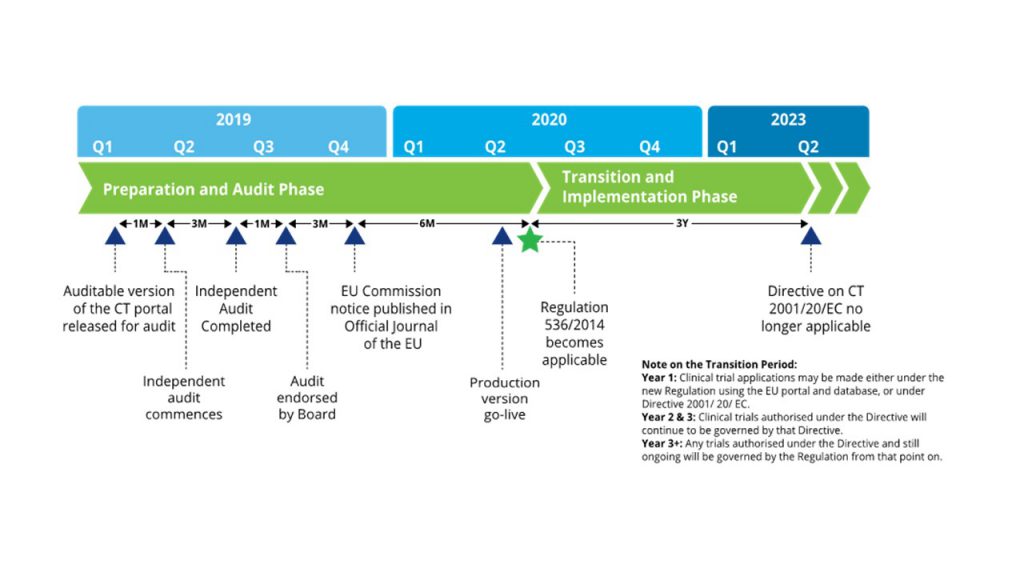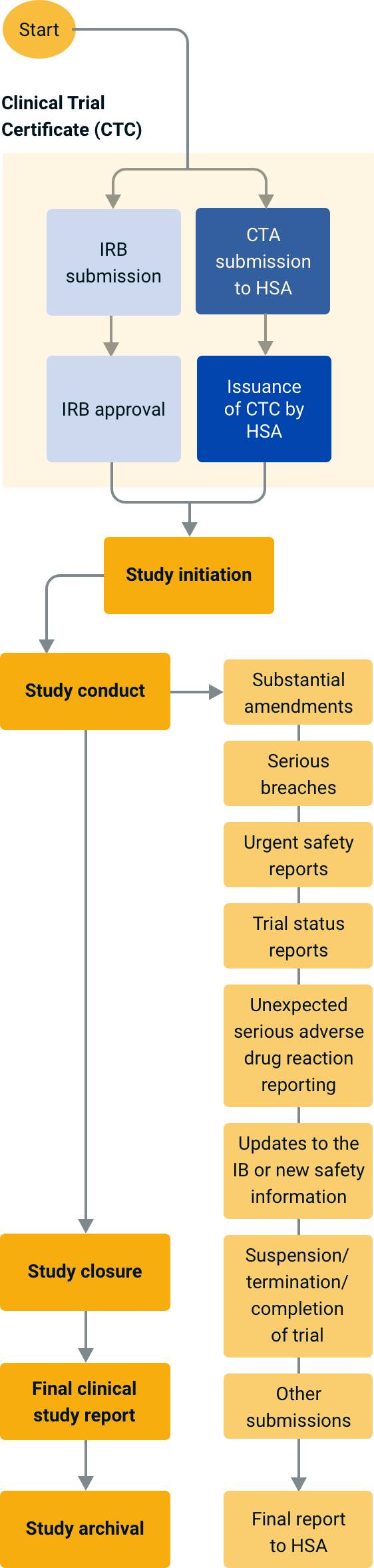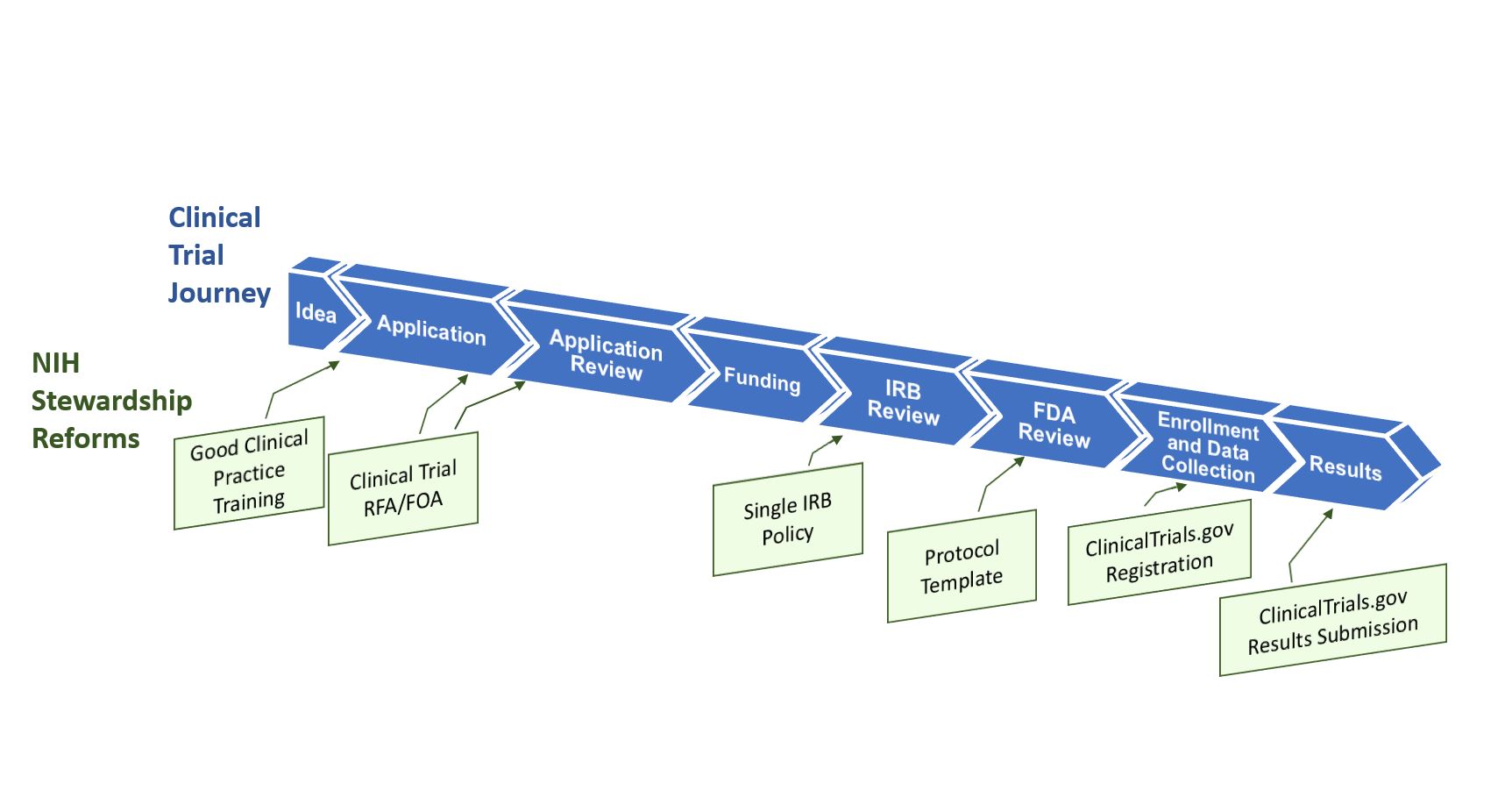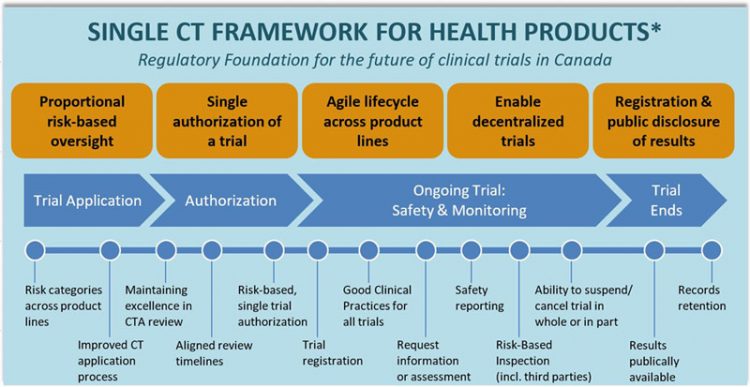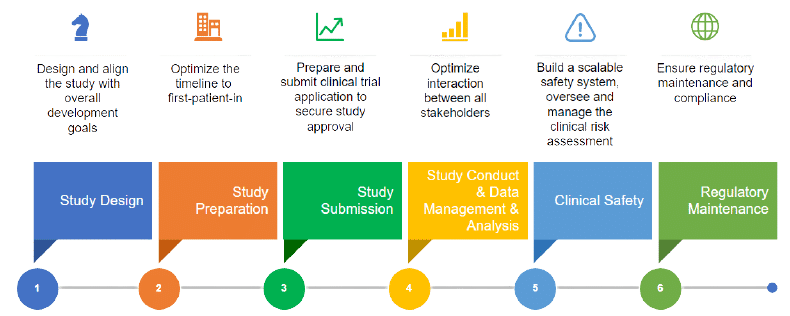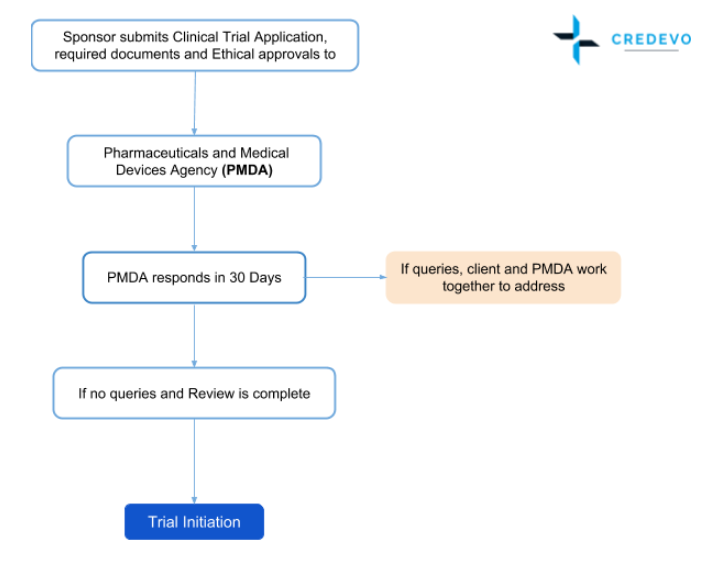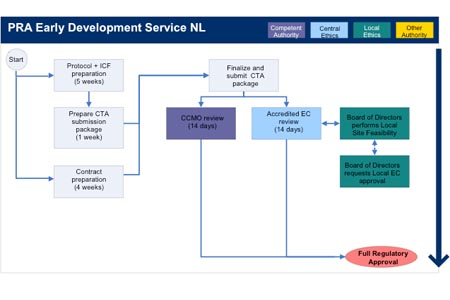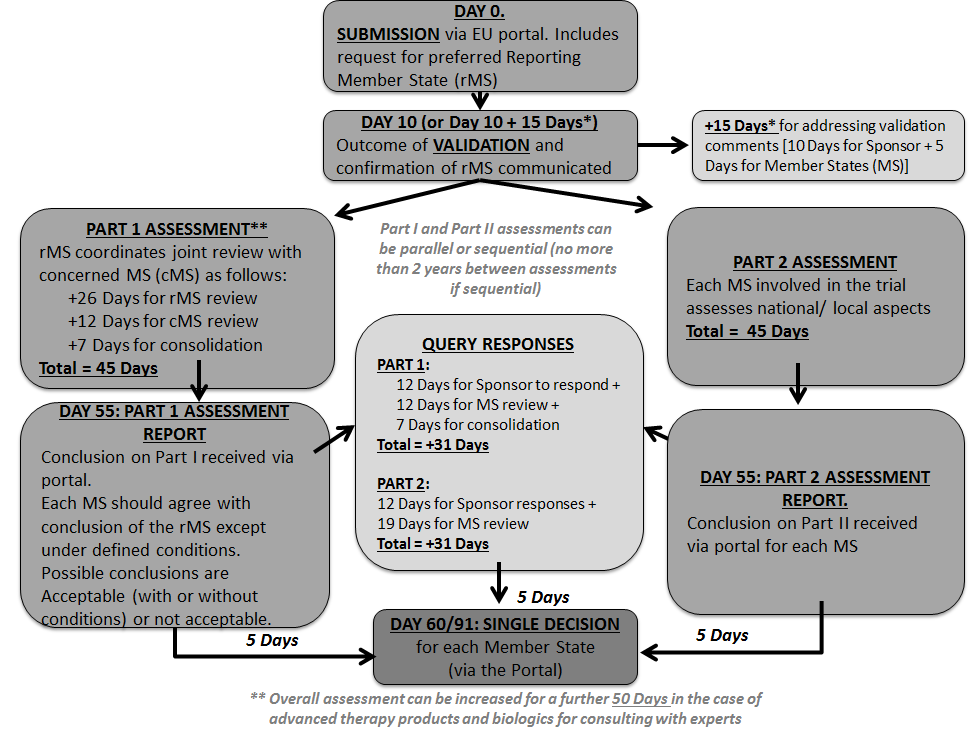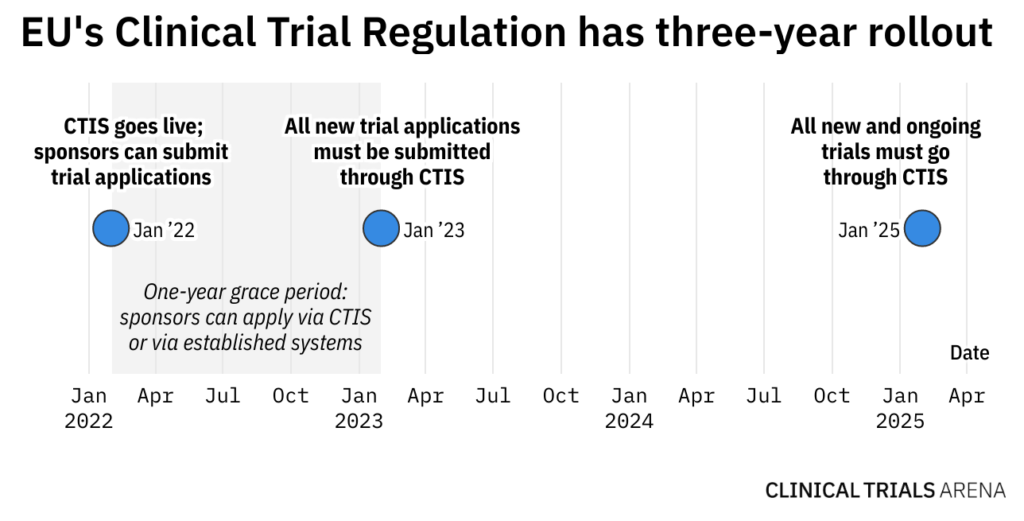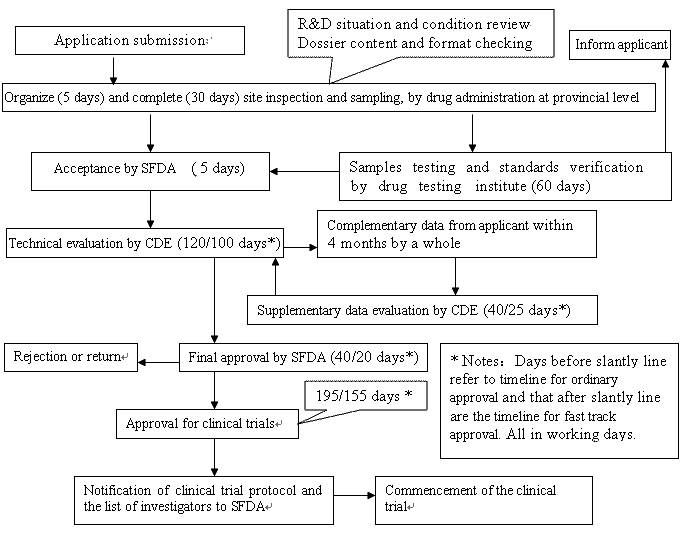
Application and approval procedure for clinical trials-China FDA,SFDA,CFDA,MOH,MOA,AQSIQ,CNCA,CIQ registration approval license for cosmetics,health food supplement,medical device,IVD,drug,infant milk powder,dairy,pet food ,disinfectant etc.

Application of clinical trial inclusion criteria to clinical practice patients to quantify the burden of CNS metastases on health-related quality of life and healthcare resource use in patients with NSCLC - Lung

Regulatory Affairs 101: Introduction to Investigational New Drug Applications and Clinical Trial Applications - Chiodin - 2019 - Clinical and Translational Science - Wiley Online Library

Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India
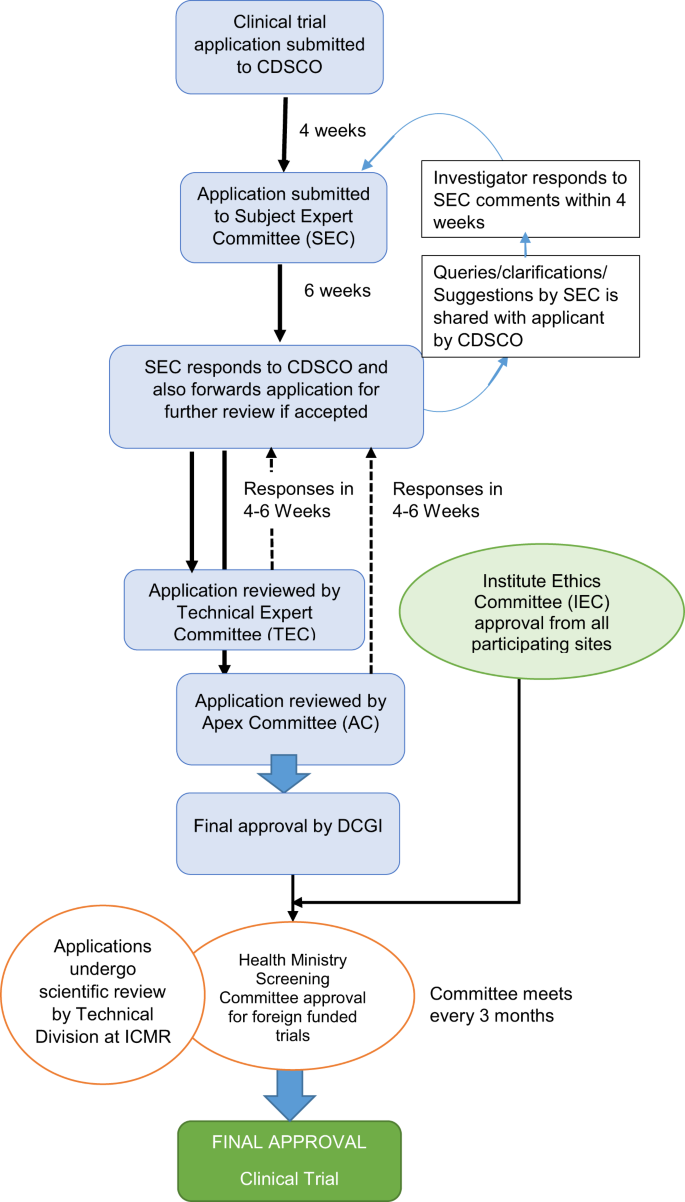
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology

Indicative clinical trial application review process in India, which... | Download Scientific Diagram

Proposed clinical trial application dossier. AMPs auxiliary medicinal... | Download Scientific Diagram